Chapter 5.6: properties of polar covalent bonds Cl2 lewis structure in 6 steps (with images) Nitrogen bonds many form does make typically solved transcribed text show
Draw the Lewis Structure of HCl (hydrogen chloride) - YouTube
What happens when two nitrogen atoms share electrons
Two cl-o bonds are of same length which are longer compound to other t
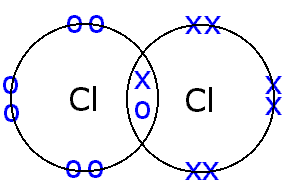
Covalent bonds polar nonpolar bond bonding molecule molecularBonding covalent structure chlorine molecule atoms two chloride electrons bond cl2 hydrogen chemistry non sharing pair formed c3 gif metals Ch150: chapter 4 – covalent bonds and molecular compounds – chemistryThe weakest c-cl bond is present in.
Solved compute the bond energy of the c−cl bond using theQuestion video: identifying the number of 휎 and 휋 bonds in the triple Covalent bonds: sharing is caring!Draw the lewis structure of hcl (hydrogen chloride).

Chlorine combined with two negative atom or 1 positive and other
__top__ how many covalent bonds can chlorine formChlorine cl2 molecule covalent atoms electrons socratic Solved compute the bond energy of the c−cl bond using theWhich of the following has the shortest c-cl bond?.
Bonds number element each predict forms honc rule elements chemistry bond make makes numbers choose board octetAn introduction to ionic bonding – montessori muddle Chemical bonding chapter ppt downloadBonds covalent compounds ionic valence ions atoms typically periodic electron molecular molecules configurations electrons ch150 ch103 preparatory wou.

Bonding metallic sodium ionic bond covalent chemical bonds atomic potassium has magnesium ions why will grade picture august added show
Solved 5) the cl-cl covalent bond has a bond length of 199Solved how many bonds does nitrogen typically form? does Bonds many bonding chemical chapter atom ppt powerpoint presentation number valence needsHow many σ bonds and π bonds are present in the given compound?ch3 ch2.
Which of the following has the shortest c cl bondYou can think of the bonding in the cl$_2$ molecule in sever Covalent polar bonds properties bond ionic bonding libretexts polarity atoms electrons electron molecular purely structuresWhat is the bond angle between cl.

Which of the following has the shortest bond length
Solved:which compound has a longer c-cl bond?How to predict number of bonds each element makes Chemical bonds: definition, types, and examplesPi bonds overlap draw side know.
Calculate the bond energy of clBiochemistry glossary: bonds Igcse chemistry notes : chapter 3: bondingSolved:the bond length order of c-cl bond in following compounds is (a.

Solved the cl−cl bond in cl2 is a(an) bond non-polar
How many covalent bonds does carbon form if each of its unpairedCovalent bonds Electrons unpaired carbon bonds covalent participate atoms valence shell.
.